K-Regio MitoFit
- mitochondria fit & well
2015-05-01 to 2018-10-31
Diagnosis of mitochondrial functions and mitochondrial injuries
- Diagnosis of mitochondrial functions and mitochondrial injuries represents a current challenge for biomedical research and development. We address these challenges by introducing and extending the approach of cell ergometry as the state-of-the-art functional test of mitochondrial competence. MitoFit establishes an inter-university Network in Mitochondrial Physiology and Comprehensive Exercise Testing, supporting translational research on mitochondrial competence in close collaboration with the local industry and other networks, e.g. Oncotyrol. Mitochondrial fitness will play an increasing role in therapeutic and preventive medicine, with exercise and caloric balance providing the most effective measures for the reduction of several age-related health risks and degenerative diseases. MitoFit is a gateway and milestone to better diagnose, support and treat patients in our modern, rapidly aging society.
MitoFit - hot topics
- MitoFit quality control - new on MitoPedia
- » Making a molecular map of exercise - NIH Common Fund
Concept
- A physically active and nutritionally healthy life style induces regulatory mechanisms that maintain aerobic performance and support functional mitochondrial competence. Even in advancing age, exercise increases aerobic fitness and maintains muscle mitochondrial density above the sedentary benchmark, contributing to the high quality of life characteristic of alpine regions such as Tyrol. Functional mitochondrial competence and aerobic fitness reduce several age-related health risks, including cardiovascular and degenerative diseases, such as type 2 diabetes, neurodegenerative diseases (Alzheimer’s, Parkinson’s, Huntington’s), and several types of cancer. As a result of a sedentary life style and progression to obesity, the decline of muscle mitochondrial capacity may contribute to accelerated aging processes and development of degenerative diseases. Diagnostic evaluation of functional mitochondrial competence requires scientific and technological mitochondrial competence. MitoFit fit&well will combine the specialized scientific focus on mitochondrial research and exercise testing with the expertise of local companies of world-wide leadership in this expanding field. Beyond the added value emanating from the combination and coordination of these diagnostic elements, the consortium will collaborate on the implementation of refined and novel diagnostic protocols as a milestone towards an internationally recognized center of excellence, with an explicit focus on sport and clinical perspectives to address mitochondrial health and disease.
Objectives
- MitoFit combines the expertise of experienced partners in a unique corporate framework. MitoFit will establish a research and diagnostic laboratory network to form a Centre of Excellence in Mitochondrial Physiology, Pathology, Comprehensive Exercise Testing and Genetic Fingerprinting. Diagnostic analyses include cardiopulmonary (VO2max; CPX) and metabolic (31P-NMR; Ergospect) exercise testing, evaluation of mitochondrial performance (high-resolution respirometry; Oroboros Instruments), and genetic screening (mtDNA haplotypes, relevant nuclear-encoded genes). Refined state-of-the-art diagnostic protocols are extended and evaluated in a training programme under acute high-altitude hypoxia versus normoxic conditions with healthy control subjects. Exposure to acute hypoxia represents a typical condition for millions of individuals annually skiing or mountaineering in Austria and particularly in Tyrol. Comprehensive mitochondrial and exercise testing protocols will be developed and validated in these studies. Case studies will translate the scientific expertise into the clinical setting. As a result, MitoFit will offer and apply these diagnostic services for (i) monitoring of training programmes in competitive athletes, (ii) life style analyses of the general population, (iii) exercise interventions for preventive and therapeutic medicine, and (iv) clinical diagnosis of individual patients.
Technological and scientific innovation
- Diagnosis of mitochondrial function has advanced to a new technological level by the introduction of high-resolution respirometry, and on a scientific level of innovation by multiple substrate-uncoupler-inhibitor titration protocols with permeabilized fibres from small muscle biopsies. Nevertheless, compared to the small needle biopsy, functional mt-diagnosis on far less invasive blood samples (thrombocytes) might provide a new perspective for large-scale screening of individual athletes, extended cohorts in clinical studies, and application for individual patients. Simultaneous measurement of mitochondrial respiration and additional parameters (mt-membrane potential by ion selective electrodes, hydrogen peroxide production by spectrofluorimetry coupled to the O2k) requires the development of new diagnostic protocols that can be applied to permeabilized muscle fibres and/or intact and permeabilized thrombocytes. Although conventional cardiopulmonary exercise testing (CPX) provides insights into the coupling of external to cellular respiration, it mirrors insufficiently the metabolic processes in the contracting and recovering muscle. Thus, combining CPX and techniques assessing bioenergetic processes in the working muscles will undoubtedly increase the conclusiveness of exercise testing in athletes and patients. These diagnostic MultiSensor protocols in combination with comprehensive exercise testing and genetic fingerprinting will generate an innovative repertoire of diagnostic tests that can be offered in the context of sports and mitochondrial diseases.
From MitoCom to MitoFit – 2020 m and above
- Join those who seek the special recreational and adventurous experience with physical exercise in the Alps. Your exercise is the gateway to your mitochondrial fitness.
- MitoFit evolves as a unique network of practitioners in the high-end sector of recreation, sport and protective medicine, in collaboration with experts on mitochondrial physiology and mitochondrial medicine.
- We all know that feeling of being well from head to toe – or from muscle to brain – up in the mountains. Scientists want to get to the basis of this wellbeing, to gain insights into invisible molecular functions: is it caused by the ’good air‘ we breathe, even though air gets very thin high up in the Alps? How do the lungs, blood, heart, brain, arms and legs – all organs and all somatic cells – react to altitude and relaxation in nature, to challenges in sport or to a short period of life on a mountain pasture?
- Mitochondria are responsible for the energy supply in human and animal tissues; this is cell ergometry, cell respiration – mitochondrial oxygen consumption. With the Oroboros O2k, we determine energy turnover of carbohydrates and fatty acids and quantify the efficiency of mitochondria in maintaining the vital body functions. In order to determine cellular fitness, parameters for oxidative stress and stress resistance are measured along with mitochondrial oxygen consumption. Biomedical research all over the world applies this method for the diagnosis of mitochondrial competence, especially in sports medicine and gerontology. The O2k, developed by the company Oroboros Instruments, is the world-wide leading instrument for diagnosing the aerobic capacity of mitochondria with the help of small samples of tissue or blood. Oroboros Instruments is located in Innsbruck, Austria. In the Mitochondrial Research Laboratory, the company tests the newest developments and develops standard protocols in cell ergometry.
- The logo of MitoCom Tyrol (now transformed to MitoFit) is based on MiPArt by Odra Noel. Further details: »MitoCom O2k-Fluorometer.
Impact on life style: Tyrol – a heart for sport
- Mitochondrial knowledge will, with its innovative approach, arouse a new awareness of preventive medicine, to which a physically active and calorie-balanced lifestyle can contribute significantly. Of particular importance is “healthy aging”, a generally improved quality of life and a reduced risk of a multiplicity of degenerative diseases influenced by the mitochondria: e.g. obesity, diabetes 2, various forms of cancer and dementias. Mitochondrial competence will be made a reality to a larger proportion of the population and the regional identity strengthened both amongst our own population and amongst tourists through the offer of exercise-orientated leisure packages in the Alpine landscape of Tyrol.
- Linkages are being created by MitoFit between research and clinical/practical needs with regard to the very recently established Olympic centre at the Institute of Sport Science of the LFU in Innsbruck and the Altitude Training Centre in Kühtai near Innsbruck leading to optimization of diagnostic exercise testing and training strategies. Importantly, this project seeks to promote research on exercise effects at moderate altitude. Tyrol represents a unique location which is visited by several millions of skiers and mountaineers, annually. On the one hand, regular physical activity and probably also exposures to moderate altitudes up to about 2500 m may contribute to well-being and longevity. On the other hand, however, unaccustomed exercise and acute altitude exposure may trigger serious cardiovascular adverse events in subjects at risk. This initiative should contribute to a better understanding of mechanisms responsible for beneficial and adverse effects supporting the development of appropriate preventive measures.
- Appreciation by politicians and communication with an aspirational general public are essential to benefit from world-class research, for comprehensive mitochondrial and exercise testing and implementation of exercise as a medicine, and the development of a scientific foundation that may allow us to translate the complex diagnostic results into a patient-related mitochondrial score. The MitoFit network will emphasize the connections to international mitochondrial networks, for exchange of expertise and standardization of diagnostic approaches, in a joint effort to helping mitochondrial patients, preventing numerous diseases and making a difference to society.
See also
- K-Regio MitoFit is the next step following the K-Regio project MitoCom (mitochondrial competence; O2k-Fluorometer), extending our world-wide technological leadership in mitochondrial respirometry with the development of a quality control system, MitoFit-Knowledge Management Platform and applications in preventive medicine (K-Regio project MitoFit).
- Health tourism: http://www.tt.com/wirtschaft/7750785-91/mehr-gesundheit-f%C3%BCr-tirol-urlauber.csp
- Sport science in focus
The project MitoFit highlights the benefits of mitochondrial fitness
* English - »German
MitoFit
MitoFit in health and preventive medicine
- Physical inactivity is one of the greatest drivers of metabolic dysregulation and preventable diseases in modern societies, and has an impact on ageing populations in particular. It is becoming increasingly apparent that high and robust mitochondrial capacities are central for sustained metabolic health throughout life. Active lifestyles increase mitochondrial capacities and reduce the risk of degenerative diseases. Despite the clear role of high and efficient mitochondrial capacities in preventing metabolic diseases, promoting health and improving life quality in ageing populations, there is currently no regimented, quantitative system, or database organised to routinely test, compare and monitor mitochondrial capacities within individuals, populations, or among populations. The Oroboros O2k - established in 49 countries - is the only instrument with sufficient stability and reliability for quantitative high-resolution respirometry (HRR). This world-leading technology offers the unique potential for developing the quality control tools required to establish the O2k as a calibrated and standardised in vitro diagnostic device. Overall, we aim to gain a CE Mark and FDA approval for the O2k, following the innovations on the level of a quality control system, and to transform this high-resolution research instrument into an essential medical device to monitor metabolic health – maintaining mitochondria fit and well to live a healthier, longer life.
K-Regio project MitoFit
- The K-Regio project MitoFit aims at developing novel laboratory standards and diagnostic monitoring of a mitochondrial fitness score. MitoFit will provide a signature to stimulate health tourism in the Alpine setting, introducing a scientific perspective on the benefits of mitochondrial fitness ranging from outdoor sports to quality of life and healthy ageing supported by physiotherapy.
- Applicant: Medical University of Innsbruck, A.Univ.-Prof. Dr. Erich Gnaiger
- Application: 2015-04-24; Granted: 2015-09-16
- Project duration: 2015-05-01 to 2018-04-30, extended to 2018-10-31
- Total budget: € 1.26 Mill; financial support by Fonds Tiroler Zukunftsstiftung (LGBl. Nr. 88/1997) € 0.89 Mill.
Concept
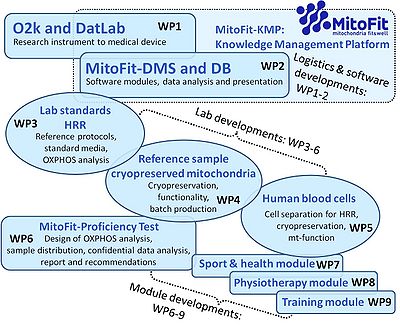
- MitoFit will provide a unique opportunity within fields of recreation, sports, wellness, lifestyle monitoring and preventive medicine programs. Collaborations with established regional initiatives will leverage on current research initiatives and established expertise in the measurement of mitochondrial function and competence. This will translate to tangible outcomes for the MitoFit concept as a marketable health, sports performance and medical diagnostic device. The establishment of the MitoFit-Knowledge Management Platform with a database reposited in the MitoFit Centre of Excellence (Fig. 1) will also provide a keystone to support the leadership of the mitochondrial monitoring instruments developed in translational research on mitochondrial competence (e.g. previous K-Regio project MitoCom) in close collaboration with local industrial partners.
- The O2k-Core and O2k-Fluorometer, which are the gold standard for generating reliable quantitative respirometric data, will be the instruments of choice to build the MitoFit database. Standardized protocols developed by the MitoFit project will be streamlined for applications of the K-Regio project MitoFit. This will lead to a significant expansion of the world-wide market for the O2k, and drive the transition of the O2k from a well established specialized research tool to a standard health and biomedical instrument.
Objectives
- The objective is to achieve the transformation for the O2k from a research instrument to a medical device. The O2k is a high-end product for measuring mitochondrial function by high-resolution respirometry and is currently distributed in 49 countries world-wide. In an increasing number of cases, we are faced with demands on CE, FDA or equivalent approval for clinical diagnosis. Presently, there is no other instrument available on the market which meets such requirements. While continuing our strategy on Open innovation to maintain our technological leadership in mitochondrial respiratory research, the objective of the present project is to initiate an additional dimension of Strategic innovation towards standardization and comprehensive quality control as required for a medical device.
Technological and scientific innovation
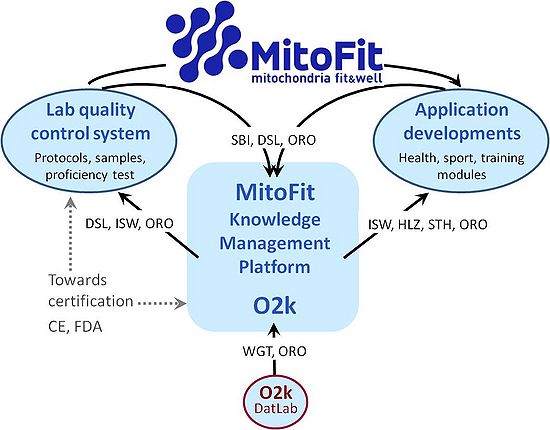
- The MitoFit-Knowledge Management Platform (KMP)
- The MitoFit KMP will be developed as an integral component of the O2k-technology. It will provide the first standardized measures to link human mitochondrial health to physiome data (heart rate, BMI, blood pressure, blood chemistry, VO2max). The technological ‘sole source’ status will be systematically secured. An increasing amount of respirometric data on mitochondrial physiology is generated world-wide, enlarging the theoretically available data base at an increasing speed. However, due to lacking standards as to how results are reported, most published data are not available for quantitative meta-analysis.
- Even within research groups, data from different projects have to be tediously re-analyzed for quantitative comparison. By extending O2k applications to monitoring personal mitochondrial fitness and diagnosing an increasing range of health conditions, the development of a knowledge management platform will become even more important and mandatory for considerations on CE and FDA approval. The design of the MitoFit-Data Management System will provide the conceptual framework for strategic innovations related to a quality control system for standardization of instrumental quality tests and mitochondrial reference protocols, establishment of reference samples for proficiency tests, and guidelines for data analysis and presentation. Based on a collection of data obtained through this platform, users will be able to search from pre-defined fields or through full-text search for data, documents and a variety of areas of interest. In combination with web-based collaboration tools implemented in the software, the platform will both broaden the actually available data base for all researchers and stimulate the development of research networks.
- The MitoFit-Knowledge Management Platform (KMP)
- Reference sample of cryopreserved mitochondria
- The development of a reference sample for respirometry will provide enormous benefits for scientific research and open up new perspectives on clinical applications, enabling a presently unattainable level of quality control in respiratory studies which will (1) increase the scientific potential of on-going and newly initiated mitochondrial studies in basic and applied research. (2) The reference sample will provide the possibility for quality control of quantitative data for direct comparison and evaluation of functional mitochondrial diagnosis on patients monitored over the timecourse of their disease and treatment, and diagnosed by different clinical laboratories. (3) A reference sample can contribute significantly to the substitution and reduction of experiments with sacrificed animals, by (a) replacing animal tissue used for training and routine quality control, or (b) for methodological development, and (c) reducing the number of artefacts in experiments with animal or human tissue by detecting methodological deficiencies in routine quality control using the reference sample.
- Reference sample of cryopreserved mitochondria
- Cryopreservation of human blood cells
- Obtaining tissue biopsies for the study of mitochondrial function is an invasive approach that requires a clinician to obtain the samples under strictly sterile conditions and that usually goes along with the need to immediately use the samples for measurements. The possibility to measure mitochondrial function in human blood cells will help to overcome these limitations by allowing a far less invasive sampling procedure applicable by anyone proficient in blood drawing and by enabling the collection and storage of samples for later measurements and analysis. It will also allow the repeated sampling of the same individual, as the burden of sampling is dramatically reduced. Altogether, the advantages of using cryopreserved human blood cells for respirometry will immensely widen the applicability of respirometry for the study of human physiology and will provide a significant stimulus for the entire research field.
- Cryopreservation of human blood cells
- MitoFit proficiency test
- A proficiency ring test, probing the performance of participating laboratories, will be applied using the reference samples developed in a previous step. Measuring the respiration of reference samples at two pre-defined time points and following strictly defined experimental protocols, it will be possible for the first time to evaluate and compare the proficiency of different labs and to assess the reproducibility of measurements conducted, thereby having a measure of quality control at hand. Based on the outcome of the test, individual labs may set adequate steps to improve their performance and to develop lab SOPs supporting compliance with defined standard requirements.
- MitoFit proficiency test
Current state-of-the-art
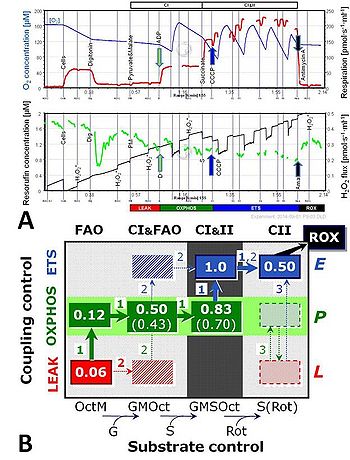
A: Combined determination of oxygen consumption and H2O2 flux by O2k-Fluorometry in permeabilized HEK cells. Substrate and coupling states are shown in the upper and lower bars. Substrate states are limited to CI- and CI&II-linked respiration. From Makrecka- Kuka et al (2015).
B: Coupling/substrate control diagrams for experimental protocol design, including multiple substrate states for fatty acid oxidation, FAO (octanoylcarnitine & malate, OctM), CI-linked (addition of glutamate, G), CI&II-linked (addition of succinate, S), and CII-linked (addition of rotenone, Rot) respiration. Flux control ratios are normalized relative to ETS capacity with convergent CI&II (&FAO) electron input. Arrows 1, 2 and 3 indicate different SUIT protocols with partially overlapping respiratory states. From Gnaiger (2014).
- Knowledge Management Platform
- Many research groups are working on extensive documentation of genetic, epigenetic, transcriptomic, proteomic, lipidomic and metabolomic datasets in basic and clinical research, including mitochondrial medicine. Relevant techniques include next-generation sequencing to determine the genome (nDNA and mtDNA), methylome, and transcriptome (RNA), and mass spectrometry (proteome, lipidome, K-Regio: MitoFit 4 MitoFit www.mitofit.org metabolome). Such data are available online through various portals, some of which provide Open Access to allow researchers to use the data for addressing their specific interests and working hypotheses.
- Knowledge Management Platform
- The capacity and efficiency of energy transformation through the mitochondrial pathways of core energy metabolism represent key features of the mitochondrial phenotype, but no corresponding data base is available for mapping mitochondrial health versus disease in humans. Performance parameters of OXPHOS are not available in a consistent format for cataloguing and, therefore, are not represented in presently available databases. Quantitative comparability of data generated by different labs on OXPHOS performance is restricted due to different protocols applied for preparing experimental solutions (solution protocols) and lack of standardization of procedure protocols including instrumental performance protocols, mitochondrial and cell preparation protocols and experimental titration protocols (SUIT protocols; Fig. 2).
- With more than 280 and 150 O2k instruments sold in the EU and the US, respectively, it appears that we are not required to obtain a CE Mark or FDA clearance or approval for the O2k. However, this status will change when extending the applications from a research instrument to a medical device intended to report personal mitochondrial fitness disposition results for a wide range of health conditions, including the propensity for degenerative and other complex diseases. According to our knowledge of the available literature, the Oroboros O2k is the only instrument on the world-wide market providing system accuracy criteria compatible with implementation of a Quality Control System for use as an In Vitro Diagnostic (IVD) device.
Workpackages
- WP1
- » The O2k from research instrument to medical device - MitoFit-Medical device
- WP1
- WP2
- » MitoFit-Knowledge Management Platform - MitoFit-KMP
- WP2
- WP3
- » Laboratory standards for high-resolution respirometry: towards a QCS - MitoFit-QCS
- WP3
- WP4
- » Reference sample: cryopreserved mitochondria - development for functional control assays - MitoFit-Reference sample
- WP4
- WP5
- » Human blood cells as study model of mitochondrial respiration and H2O2 production - MitoFit-Bloodcells
- WP5
- WP6
- » Development of the MitoFit proficiency test - MitoFit-PT
- WP6
- WP7
- » Development of a sport module: high altitude sprint training - MitoFit-Sport module
- WP7
- WP8
- » Development of a physiotherapy module: effects of physical activity on cardiovascular risk factors in middle-aged subjects - MitoFit-Physiotherapy module
- WP8
- WP9
- » Development of a training module - MitoFit-Training module
- WP9
Partners
![]() MUI: Universitätsklinik für Visceral-, Transplantations- und Thoraxchirurgie, D. Swarovski Forschungslabor, DSL (Ao. Univ.-Prof. Dr. Erich Gnaiger)
MUI: Universitätsklinik für Visceral-, Transplantations- und Thoraxchirurgie, D. Swarovski Forschungslabor, DSL (Ao. Univ.-Prof. Dr. Erich Gnaiger)
![]() LFU: Institut für Sportwissenschaften, ISW (Univ.-Prof. DDr. Martin Burtscher, Verena Menz, Dr. Hannes Gatterer)
LFU: Institut für Sportwissenschaften, ISW (Univ.-Prof. DDr. Martin Burtscher, Verena Menz, Dr. Hannes Gatterer)
![]() SME: Oroboros Instruments GmbH, Innsbruck, ORO (Dr. Erich Gnaiger, Mag.a. Verena Laner)
SME: Oroboros Instruments GmbH, Innsbruck, ORO (Dr. Erich Gnaiger, Mag.a. Verena Laner)
![]() SME: WGT-Elektronik GmbH & Co KG, Kolsass, Tyrol, WGT (Philipp Gradl)
SME: WGT-Elektronik GmbH & Co KG, Kolsass, Tyrol, WGT (Philipp Gradl)
![]() SME: Höhenleistungszentrum Betriebsg.m.b.H., Innsbruck, HLZ (Geschäftsführer Mag. Andreas Brix, Robert Valentini)
SME: Höhenleistungszentrum Betriebsg.m.b.H., Innsbruck, HLZ (Geschäftsführer Mag. Andreas Brix, Robert Valentini)
![]() SME: Sporttherapie Mag. Huber GmbH, Innsbruck, STH (Mag. Reinhard Huber)
SME: Sporttherapie Mag. Huber GmbH, Innsbruck, STH (Mag. Reinhard Huber)
MUI = Medical University Innsbruck; LFU = Leopold-Franzens-University Innsbruck; SME = Small and medium-sized enterprise
External consultants
- MUI: Department für Medizinische Statistik, Informatik und Gesundheitsökonomie, MSIG (Univ.-Doz. Dr. Georg Göbel)
- software security networks - ssn (Lukas Gradl)
- fancy tree films (Johannes Aitzetmüller)
- en.co.tec Schmid KG, Technisches Büro für Elektrotechnik (Dipl.- Ing. Martin Schmid)
International strategic partners - Europe
- Garcia-Roves Pablo Maria, PhD Assoc. Prof.: Faculty of Medicine, University of Barcelona, ES
- Karabatsiakis Alexander, PhD: Clinical and Biological Psychology, University of Ulm, DE
- Carsten Lundby, PhD Prof.: Institute of Physiology, University of Zurich, CH
- Sumbalova Zuzana, PhD: Pharmacobiochemical Laboratory, Faculty of Medicine, Comenius University, Bratislava, SK
International strategic partners - overseas
- Goodpaster Bret H, Prof. Dr: Translational Research Institute for Metabolism and Diabetes, Florida Hospital, Sanford/Burnham Institute, Orlando, FL, US
- Hickey Anthony, PhD: School of Biological Sciences, The University of Auckland, NZ
- Molina Anthony JA, PhD: J. Paul Sticht Center on Aging, Dept Internal Medicine, Wake Forest School of Medicine, Winston-Salem, NC, US
- Neufer P Darrell, Prof. Dr. & Brown David A, PhD: Dept of Exercise and Sport Science & Brody School of Medicine, East Carolina University, Greenville, NC, US
- Ojuka Edward O, Prof. Dr. PhD: Institute of South Africa Newlands, ESSM UCT Dept of Human Biology Sports Science, University of Cape Town, ZA
Becoming ‘MitoFit’ – a trend towards a new lifestyle?
- With its innovative approach, MitoFit arouses a new awareness of preventive medicine, to which a physically active and calorie-balanced lifestyle can contribute significantly. Of particular importance is “healthy aging”, a generally improved quality of life and a reduced risk of a multiplicity of degenerative diseases influenced by the mitochondria: e.g. obesity, diabetes II, various forms of cancer and dementias. Mitochondrial competence will be made a reality to a larger proportion of the population and the regional identity strengthened both amongst our own population and amongst tourists through the offer of exercise-orientated leisure packages in the Alpine landscape of Tyrol.
- Making our mitochondria fit – MitoFit and well – may strengthen the trend to a less sedentary, healthier and balanced lifestyle, supported by the growing evidence of the central role of mitochondrial function for an improved and prolonged quality of life.
Related projects
- COST Action » MitoEAGLE
- Tiroler Innovationsförderung » O2k-Innovation (Feb 2015 - Jan 2017)
- K-Regio MitoCom: » MitoCom (completed 2014)
MitoFit project news
2016
- 003. 2016-05-06:
- Dear colleague,
- 1st MitoFit Science Camp, Kuehtai (close to Innsbruck) – 2016, July 07 to 13.
- Programme update: » MitoFit Science Camp 2016 Kuehtai AT
- Abstract deadline: extended to May 27.
- Early bird registration until May 15: » MitoFit SC 2016 Registration
- 002. 2016-03-30:
- Dear colleague,
- We are happy to inform you that registration for the 1st MitoFit Science Camp is now open. Immediately following EBEC2016 (July 2-7), the MitoFit Science Camp will take place in Kuehtai (close to Innsbruck), Austria from July 7th to 13th.
- High-level training in mitochondrial respiratory physiology,
- Internationally renowned speakers – interlaboratory networking,
- High-resolution respirometry - mitochondrial performance testing,
- Discussions on fundamentals of quality control – MitoFit proficiency test
- Find out more: » MitoFit Science Camp 2016 Kuehtai AT
- Register now: » MitoFit SC 2016 Registration
- We are looking forward to seeing you highly MitoFit at 2,020 m in Kuehtai,
- 001. 2016-03-08:
- We are delighted to inform the mitochondrial community about the COST Action 'Mitochondrial mapping: Evolution - Age - Gender - Lifestyle - Environment (MitoEAGLE)', which is going to be funded for four years:
- » MitoEAGLE
- Researchers from COST Member Countries and Cooperating States can join the Action. For more information (including Near Neighbour Countries and International Partner Countries) and modalities for joining, see:
- » Join the COST Action CA15203 MitoEAGLE
2015
- 020. 2015-12-22: Quote from Pedersen and Saltin (2015): ’.. it is now time that the health systems create the necessary infrastructure to ensure that supervised exercise can be prescribed as medicine.’ » Pedersen 2015 Scand J Med Sci Sports
- MitoFit hot topics:
- MitoFit Workshop on blood cell respirometry for mitochondrial research and diagnostic applications. Innsbruck, 2016-Jan-08:
- More details » MitoFit Workshop Blood Cells 2016-01
- Making a molecular map of exercise - NIH Common Fund:
- As the end of the year approaches, we thank you for your positive feedback and cooperation, sending you season's greetings and all the best for a happy, successful, MitoFit and generally healthy New Year.
- 019. 2015-12-10: From insulin-resistant obese to MitoFit – with aerobic exercise training: » Konopka_2015_Diabetes
- 018. 2015-11-25: Performance tests on skeletal muscle mitochondria of the Inuit haplogroup reveal fitness information beyond the uncoupling hypothesis for adaptations to the arctic climate: » Gnaiger 2015 Scand J Med Sci Sports
- 017. 2015-11-16: MitoFit in the brain » Burtscher 2015 Mitochondrion
- 016. 2015-10-20: In an effort to examine the comparability of data generated by different laboratories and across various studies, we are taking note of commonly used fatty acids being utilized to examine beta-oxidation.
- Therefore, we would like to ask you the following:
- What is your experience with examining fatty acid oxidation in permeabilized muscle fibres?
- Which fatty acids do you use?
- A) Octanoylcarnitine
- B) Palmitylcarnitine
- C) Other (please specify)
- The results of this survey will be summarized in the O2k-Network discussion forum: » O2k-Network discussion forum
- 015. 2015-09-24: The MitoFit project was approved by the Tyrolean Government with a budget of € 1.3 Mill. Our main aim is the development of novel laboratory standards and diagnostic monitoring of a mitochondrial fitness score. The Universities of Innsbruck and industry partners collaborate with international strategic partners in this ambitious programme:
- MitoFit Training Camp 2016 Kuehtai AT: Immediately following EBEC 2016 organized by Paolo Bernardi in Riva del Garda, the first ‘MitoFit Training Camp’ will take place on 2016 July 7-13, at the High Altitude Training Center Kühtai at an altitude of 2020 m. The MitoFit Training Camp will provide a unique opportunity to receive first-hand introductions to state-of-the-art diagnostic monitoring of mitochondrial respiratory function. A wide range of areas will be covered on protective medicine, exercise physiology, mitochondrial pharmacology, aging, to comparative mitochondrial physiology (cell types, tissues, species).
- » MitoFit Science Camp 2016 Kuehtai AT
- 014. 2015-09-02: Defining mt-hypoxia at cytochrome redox steady-states: » Harrison 2015 J Appl Physiol
- Open Innovation: the O2k-Innovation is not yet in series production, but some special innovations are now available:
- » TIP2k-Module with 200 µL (new) or 500 µl syringes, for steady-state hypoxia and more.
- » Oroboros racks make experiments with SUIT protocols more convenient.
- 013. 2015-07-31: To the heart of the Amazon - Phenotypic mitochondrial respiratory control differs widely from mouse to man. Does this mean that these hearts have different mitochondrial fitness? What can we learn from comparative mitochondrial physiology? Find out more: » MiPNet20.12 IOC103 Manaus
- The Oroboros-team sends you best wishes for a MitoFit Summer
- from this year's KrassFit/XLETIX challenge in Kühtai (Tyrol, Austria). » Oroboros MitoFit team KrassFit2015 Kuehtai
- joining with Lemawork Ketema (Ethiopia): Wings for Life World Run champion 2014 and 2015, running 79.9 km (49.9 miles) and this year's Rio de Janeiro Marathon runner #2. » Becoming MitoFit
- Summer holiday break during August 2015.
- 012. 2015-07-16: High fat/high fructose and modulation of hepatic mitochondrial function. » Aroor 2015 Diabetes
- 011. 2015-07-07: MitoFit quality above quantity in resistance training. » Porter 2015 Med Sci Sports Exerc
- 010. 2015-07-01: Light in the powerhouse of the cells - SUIT protocols for measurement of respiration and H2O2 production: » Makrecka-Kuka 2015 Biomolecules
- 009. 2015-06-23: Fit and fat - the good, the bad, and the ugly. » Schoettl 2015 Endocrinology
- 008. 2015-06-16: HIT and fit: 2-weeks of high intensity interval training. » Vincent 2015 Front Physiol
- 007. 2015-06-09: Combined training and good news for the aging population. » Irving 2015 J Clin Endocrinol Metab
- 006. 2015-06-02: Aggression and mitochondrial fitness taken from a friend: acquisition of mtDNA from host cells. » Tan 2015 Cell Metab - This is another exciting publication not in line with the controversial 'Warburg effect'.
- 005. 2015-05-26: Among the fittest, females are superior - but not always welcome. » Soares 2015 PLoS One
- 004. 2015-05-18: Exercise as medicine: protecting from diabetic cardiomyopathy. » Hafstad 2015 Antioxid Redox Signal
- 003. 2015-05-13: The power of the mind: the cortex as a critical determinant of muscle strength. » Clark 2014 J Neurophysiol
- 002. 2015-05-08: Depressed cellular energetics correlates with depressive states of mind: high-resolution respirometry in blood cells (PBMC). » Karabatsiakis 2014 Translational Psychiatry
- 001. 2015-05-05: Any type of exercise helps - strength and endurance training in the fight against obesity. » Benito 2015 J Appl Physiol
Feedback
- .. the mito-fit paper on the elderly was great! Anthony Hickey; O2k-Network Lab: NZ Auckland Hickey AJ (2015-06-10).
- Thank you for the update publication. It's quite inspiring. Ping Zheng (2015-06-09).
- Thanks for sending this interesting study conducted in PBMC for the bioblast group. Marcus Oliveira; O2k-Network Lab: BR Rio de Janeiro Oliveira MF (2015-05-08).
- This is the first glimpse I have seen of MitoFit, and I have to tell you it's brilliant! When we launched Team MitoCanada a few years back combined with all the recent research and correlation to general health and Mito health, it just seems like the natural progression. We are taking a new approach to mitochondrial health as well with MitoCanada and reading your webpage really confirms the need to consider the full spectrum of mitochondrial health as part of our awareness strategy. Blaine Penny, President and CEO of MitoCanada Foundation and winner of the Wings for Life World Run in Niagara Falls 2015 (2015-05-06).
Sort in ascending/descending order by a click on one of the small symbols in squares below. Default sorting: chronological. Empty fields appear first in ascending order.
| Year | Reference | Mammal and model | Tissue and cell | Stress | Diseases | |
|---|---|---|---|---|---|---|
| Bowen 2015 Eur J Heart Fail | 2015 | Bowen TS, Rolim NP, Fischer T, Baekkerud FH, Medeiros A, Werner S, Brønstad E, Rognmo O, Mangner N, Linke A, Schuler G, Silva GJ, Wisløff U, Adams V (2015) Heart failure with preserved ejection fraction induces molecular, mitochondrial, histological, and functional alterations in rat respiratory and limb skeletal muscle. Eur J Heart Fail 17:263-72. | Rat | Skeletal muscle | Cardiovascular | |
| Pedersen 2015 Scand J Med Sci Sports | 2015 | Pedersen BK, Saltin B (2015) Exercise as medicine - evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand J Med Sci Sports 25:1-72. https://doi.org/10.1111/sms.12581 | Human | Cancer Cardiovascular Diabetes Neurodegenerative Obesity Parkinson's Other | ||
| Neves 2014 J Nutr Biochem | 2014 | Neves FA, Cortez E, Bernardoa AF, Mattos ABM, Vieira AK, Malafaia TO, Thole AA, Rodrigues-Cunha ACS, Garcia-Souza EP, Sichieri R, Moura AS (2014) Heart energy metabolism impairment in Western-diet induced obese mice. J Nutr Biochem 25:50-57. | Mouse | Heart | Obesity |