Betiu 2019 MiP2019: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Abstract | {{Abstract | ||
|title=[[Image:MiPsocietyLOGO.JPG|left|90px|Mitochondrial Physiology Society|MiPsociety]] Cell-permeable succinate improves amiodarone-induced mitochondrial | |title=[[Image:MiPsocietyLOGO.JPG|left|90px|Mitochondrial Physiology Society|MiPsociety]] Cell-permeable succinate improves amiodarone-induced mitochondrial Complex I and II inhibition in human platelets. | ||
|info=[[MiP2019]] | |info=[[MiP2019]] | ||
|authors=Betiu AM, Aasander Frostner E, Ehinger J, Petrescu L, Muntean DM, Elmer E | |authors=Betiu AM, Aasander Frostner E, Ehinger J, Petrescu L, Muntean DM, Elmer E | ||
| Line 6: | Line 6: | ||
|event=MiP2019 | |event=MiP2019 | ||
|abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | |abstract=[[Image:MITOEAGLE-logo.jpg|left|100px|link=http://www.mitoglobal.org/index.php/MITOEAGLE|COST Action MitoEAGLE]] | ||
Amiodarone is one of the most commonly prescribed anti-arrhythmic drugs in the management of both supraventricular and ventricular arrhythmias [1]. Despite its beneficial properties, several adverse effects have been classically reported. Mitochondrial dysfunction has recently emerged as a major pathomechanism underlying amiodarone toxicity [2]. Studies conducted in mouse liver, hamster lung and human hepatocytes cells reported that amiodarone mitochondrial toxicity was caused by an impairment of | Amiodarone is one of the most commonly prescribed anti-arrhythmic drugs in the management of both supraventricular and ventricular arrhythmias [1]. Despite its beneficial properties, several adverse effects have been classically reported. Mitochondrial dysfunction has recently emerged as a major pathomechanism underlying amiodarone toxicity [2]. Studies conducted in mouse liver, hamster lung and human hepatocytes cells reported that amiodarone mitochondrial toxicity was caused by an impairment of Complexes I and II of respiratory chain [2, 3]. Succinate is a Complex II substrate that potentially can alleviate such metabolic dysfunction; however, it does not readily permeate the cell membrane. Recently, cell-permeable succinate prodrugs have been developed in order to bypass mitochondrial Complex I deficiency of different etiology [4]. | ||
The present study was purported to assess the effects of amiodarone on mitochondrial respiration in intact and permeabilized human platelets in the presence vs. the absence of NV118, a cell-permeable succinate prodrug. To this aim peripheral blood platelets were isolated from healthy volunteers by differential centrifugations (using K<sub>2</sub>-EDTA as anticoagulant) and exposed to increasing concentrations of amiodarone in the presence vs. absence of NV118; the compound was generously provided by NeuroVive Pharmaceutical AB (Lund, Sweden), also available as the MitoKit-CII from Oroboros Instruments GmbH, Innsbruck, Austria. Respiratory capacities of intact and digitonin-permeabilized platelets (200 x 10<sup>6</sup> cells/mL) were analyzed using the substrate-uncoupler-inhibitor titration protocol. | The present study was purported to assess the effects of amiodarone on mitochondrial respiration in intact and permeabilized human platelets in the presence vs. the absence of NV118, a cell-permeable succinate prodrug. To this aim peripheral blood platelets were isolated from healthy volunteers by differential centrifugations (using K<sub>2</sub>-EDTA as anticoagulant) and exposed to increasing concentrations of amiodarone in the presence vs. absence of NV118; the compound was generously provided by NeuroVive Pharmaceutical AB (Lund, Sweden), also available as the MitoKit-CII from Oroboros Instruments GmbH, Innsbruck, Austria. Respiratory capacities of intact and digitonin-permeabilized platelets (200 x 10<sup>6</sup> cells/mL) were analyzed using the substrate-uncoupler-inhibitor titration protocol. | ||
| Line 48: | Line 48: | ||
== References == | == References == | ||
::::#Soar J, Perkins GD, Maconochie I, Böttiger BW, Deakin CD, Sandroni C, Olasveengen TM, Wyllie J, Greif R, Lockey A, Semeraro F, Van de Voorde P, Lott C, Bossaert L, Monsieurs KG, Nolan JP (2019) European Resuscitation Council Guidelines for Resuscitation: 2018 Update - Antiarrhythmic drugs for cardiac arrest. Resuscitation. 134:99-103. | ::::# Soar J, Perkins GD, Maconochie I, Böttiger BW, Deakin CD, Sandroni C, Olasveengen TM, Wyllie J, Greif R, Lockey A, Semeraro F, Van de Voorde P, Lott C, Bossaert L, Monsieurs KG, Nolan JP (2019) European Resuscitation Council Guidelines for Resuscitation: 2018 Update - Antiarrhythmic drugs for cardiac arrest. Resuscitation. 134:99-103. | ||
::::#Ramachandran A, Visschers R, Duan L, Akakpo JY, Jaeschke, H (2018) Mitochondrial dysfunction as a mechanism of drug-induced hepatotoxicity: current understanding and future perspectives. J Clin Transl Res 4:75–100. | ::::# Ramachandran A, Visschers R, Duan L, Akakpo JY, Jaeschke, H (2018) Mitochondrial dysfunction as a mechanism of drug-induced hepatotoxicity: current understanding and future perspectives. J Clin Transl Res 4:75–100. | ||
::::#Silva Santos LF, Stolfo A, Calloni C, Salvador M (2017) Catechin and epicatechin reduce mitochondrial dysfunction and oxidative stress induced by amiodarone in human lung fibroblasts. J Arrhythm 33:220–25. | ::::# Silva Santos LF, Stolfo A, Calloni C, Salvador M (2017) Catechin and epicatechin reduce mitochondrial dysfunction and oxidative stress induced by amiodarone in human lung fibroblasts. J Arrhythm 33:220–25. | ||
::::#Ehinger JK, Piel S, Ford R, Karlsson M, Sjövall F, Frostner EÅ, Morota S, Taylor RW, Turnbull DM, Cornell C, Moss SJ, Metzsch C, Hansson MJ, Fliri H, Elmér E (2016) Cell-permeable succinate prodrugs bypass mitochondrial | ::::# Ehinger JK, Piel S, Ford R, Karlsson M, Sjövall F, Frostner EÅ, Morota S, Taylor RW, Turnbull DM, Cornell C, Moss SJ, Metzsch C, Hansson MJ, Fliri H, Elmér E (2016) Cell-permeable succinate prodrugs bypass mitochondrial Complex I deficiency. Nat Commun 7:12317. | ||
Revision as of 01:49, 9 October 2019
| Cell-permeable succinate improves amiodarone-induced mitochondrial Complex I and II inhibition in human platelets. |
Link: MiP2019
Betiu AM, Aasander Frostner E, Ehinger J, Petrescu L, Muntean DM, Elmer E (2019)
Event: MiP2019
Amiodarone is one of the most commonly prescribed anti-arrhythmic drugs in the management of both supraventricular and ventricular arrhythmias [1]. Despite its beneficial properties, several adverse effects have been classically reported. Mitochondrial dysfunction has recently emerged as a major pathomechanism underlying amiodarone toxicity [2]. Studies conducted in mouse liver, hamster lung and human hepatocytes cells reported that amiodarone mitochondrial toxicity was caused by an impairment of Complexes I and II of respiratory chain [2, 3]. Succinate is a Complex II substrate that potentially can alleviate such metabolic dysfunction; however, it does not readily permeate the cell membrane. Recently, cell-permeable succinate prodrugs have been developed in order to bypass mitochondrial Complex I deficiency of different etiology [4].
The present study was purported to assess the effects of amiodarone on mitochondrial respiration in intact and permeabilized human platelets in the presence vs. the absence of NV118, a cell-permeable succinate prodrug. To this aim peripheral blood platelets were isolated from healthy volunteers by differential centrifugations (using K2-EDTA as anticoagulant) and exposed to increasing concentrations of amiodarone in the presence vs. absence of NV118; the compound was generously provided by NeuroVive Pharmaceutical AB (Lund, Sweden), also available as the MitoKit-CII from Oroboros Instruments GmbH, Innsbruck, Austria. Respiratory capacities of intact and digitonin-permeabilized platelets (200 x 106 cells/mL) were analyzed using the substrate-uncoupler-inhibitor titration protocol.
In both intact and permeabilized cells, amiodarone elicited a dose-dependent (15-240 μM) inhibition of both NADH- and succinate-linked respiration. The addition of the cell-permeable succinate prodrug normalized the respiratory capacity at the electron transport system.
Amiodarone induced an acute inhibition of respiration in human non-cultured cells that was alleviated by cell-permeable succinate. Whether these results (drug effect and treatment response) can be seen in platelets isolated from patients chronically treated with amiodarone warrants further investigation.
• Keywords: Mitochondrial Dysfunction, Platelets, Amiodarone, Cell-permeable succinate • Bioblast editor: Plangger M, Tindle-Solomon L • O2k-Network Lab: SE Lund Elmer E, RO Timisoara Muntean DM
Labels: MiParea: Respiration, Pharmacology;toxicology Pathology: Cardiovascular
Organism: Human Tissue;cell: Platelet Preparation: Permeabilized cells, Intact cells
Coupling state: LEAK, ET
Pathway: ROX
HRR: Oxygraph-2k
Affiliations
- Bețiu AM(1), Åsander Frostner E(2,3), Ehinger J(2), Petrescu L(4), Muntean DM(1,*), Elmér E(2,3,5)
- Dept Functional Sciences - Pathophysiology, *Centre Translational Research & Systems Medicine, "Victor Babeș" Univ Medicine Pharmacy, Timisoara, Romania
- Dept Clinical Sciences, Mitochondrial Medicine, Lund Univ, Sweden
- NeuroVive Pharmaceutical AB, Medicon Village, Lund, Sweden
- Dept Cardiology, Inst Cardiovascular Diseases, "Victor Babeș" Univ Medicine Pharmacy, Timisoara, Romania
- Skane Univ Hospital, Clinical Neurophysiology, Lund, Sweden. - [email protected]
- Bețiu AM(1), Åsander Frostner E(2,3), Ehinger J(2), Petrescu L(4), Muntean DM(1,*), Elmér E(2,3,5)
Figures
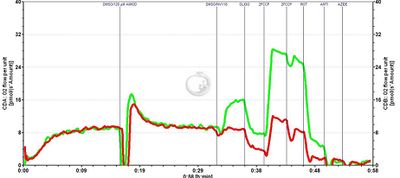
Figure 1: Oxygen consumption of intact platelets in the presence of amiodarone: the effect of succinate prodrug (NV118), in lime vs. control, in red (DMSO).
References
- Soar J, Perkins GD, Maconochie I, Böttiger BW, Deakin CD, Sandroni C, Olasveengen TM, Wyllie J, Greif R, Lockey A, Semeraro F, Van de Voorde P, Lott C, Bossaert L, Monsieurs KG, Nolan JP (2019) European Resuscitation Council Guidelines for Resuscitation: 2018 Update - Antiarrhythmic drugs for cardiac arrest. Resuscitation. 134:99-103.
- Ramachandran A, Visschers R, Duan L, Akakpo JY, Jaeschke, H (2018) Mitochondrial dysfunction as a mechanism of drug-induced hepatotoxicity: current understanding and future perspectives. J Clin Transl Res 4:75–100.
- Silva Santos LF, Stolfo A, Calloni C, Salvador M (2017) Catechin and epicatechin reduce mitochondrial dysfunction and oxidative stress induced by amiodarone in human lung fibroblasts. J Arrhythm 33:220–25.
- Ehinger JK, Piel S, Ford R, Karlsson M, Sjövall F, Frostner EÅ, Morota S, Taylor RW, Turnbull DM, Cornell C, Moss SJ, Metzsch C, Hansson MJ, Fliri H, Elmér E (2016) Cell-permeable succinate prodrugs bypass mitochondrial Complex I deficiency. Nat Commun 7:12317.