Acuna-Castroviejo 2012 Abstract Bioblast: Difference between revisions
No edit summary |
Beno Marija (talk | contribs) No edit summary |
||
| (31 intermediate revisions by 7 users not shown) | |||
| Line 1: | Line 1: | ||
{{Abstract | {{Abstract | ||
|title=Acuna-Castroviejo D (2012) Melatonin and the quality of life: | |title=Acuna-Castroviejo D (2012) Melatonin and the quality of life: the melatonin-mitochondrion connection. Mitochondr Physiol Network 17.12. | ||
|info=[[MiPNet17.12 Bioblast 2012|MiPNet17.12 Bioblast 2012 - Open Access]] | |info=[[MiPNet17.12 Bioblast 2012|MiPNet17.12 Bioblast 2012 - Open Access]] | ||
|authors=Acuna-Castroviejo D | |authors=Acuna-Castroviejo D | ||
|year=2012 | |year=2012 | ||
|event=[[Bioblast 2012]] | |event=[[Bioblast 2012]] | ||
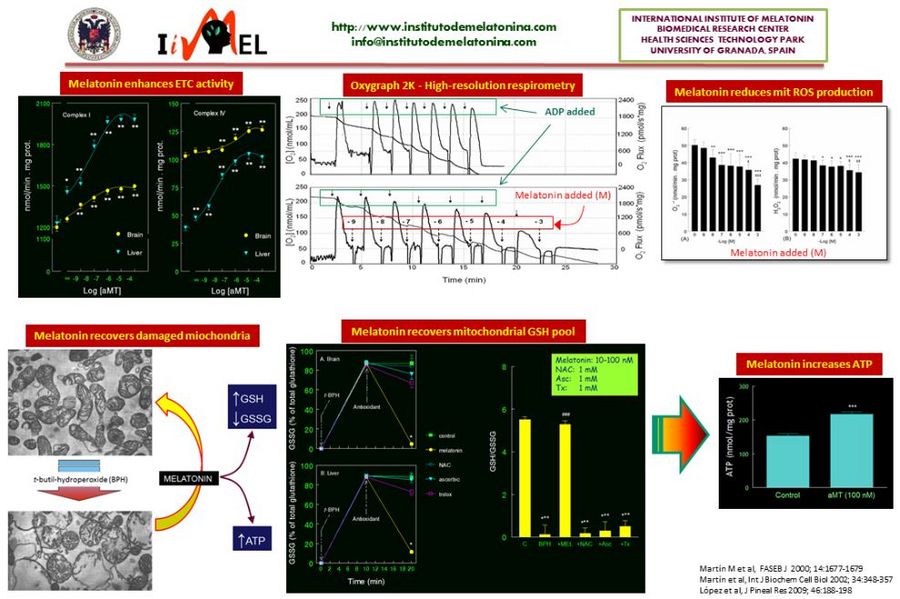
|abstract=The first relationship between melatonin and mitochondria came from histological studies showing changes in mitochondrial density and morphology after pinealectomy or melatonin administration to experimental animals. After the discovery of the antioxidant activity of melatonin in 1993, the possibility that melatonin exerts its effects on the mitochondria, the main ROS-producing organelle, was hypothesized. The first experiments demonstrated a highly efficient ability of melatonin to counteract the mitochondrial oxidative stress in vitro and in vivo. In parallel, melatonin increases the respiratory chain activity, reduces the oxygen consumption, and increases the ATP production. In some of these experiments we could demonstrated that the mitochondria take up melatonin in a time- and concentration-dependent manner. To further analyze the ability of melatonin to prevent and/or counteract mitochondrial dysfunction, different experimental models of aging and disease, including sepsis, Parkinson’s disease, and Alzheimer’s disease, was evaluated. In all of them, melatonin administration restored the full bioenergetic capacity of the mitochondria, restoring or even increasing their ATP production. Along this time, it was | |abstract=[[File:Dario.JPG|right|200px|Dario Acuna-Castroviejo]] | ||
The first relationship between melatonin and mitochondria came from histological studies showing changes in mitochondrial density and morphology after pinealectomy or melatonin administration to experimental animals. After the discovery of the antioxidant activity of melatonin in 1993, the possibility that melatonin exerts its effects on the mitochondria, the main ROS-producing organelle, was hypothesized. The first experiments demonstrated a highly efficient ability of melatonin to counteract the mitochondrial oxidative stress ''in vitro'' and ''in vivo''. In parallel, melatonin increases the respiratory chain activity, reduces the oxygen consumption, and increases the ATP production. In some of these experiments we could demonstrated that the mitochondria take up melatonin in a time- and concentration-dependent manner. To further analyze the ability of melatonin to prevent and/or counteract mitochondrial dysfunction, different experimental models of aging and disease, including sepsis, Parkinson’s disease, and Alzheimer’s disease, was evaluated. In all of them, melatonin administration restored the full bioenergetic capacity of the mitochondria, restoring or even increasing their ATP production. Along this time, it was shown that most of the tissues and organs produce melatonin independently of the pineal gland. An important feature of this extrapineal source of melatonin is that its synthetizing enzymes, AANAT and ASMT, are inducible, i.e., the cells produce melatonin when they require it for protective purposes. This melatonin does not exit to the extracellular fluid. To further analyze the dynamics of the extrapineal melatonin, we recently studied the subcellular distribution of the indoleamine in liver and brain. These studies showed that melatonin is produced in considerably higher amounts in these tissues than in the pineal gland, it is not uniformly distributed in the cell, and is mainly located in the membrane, mitochondria and nucleus. Interestingly, membrane content of melatonin increased in a dose-dependent manner after administration of melatonin to rats, but the content of the indoleamine in the nucleus and mitochondria is saturated. There are now evidences of the ability of mitochondria (and chloroplasts) to synthesize melatonin, which explains the high levels of the indoleamine in this organelle. The phylogenetic origins of the mitochondria, and the presence of melatonin in ancient one cell organisms, speak in favor of a melatonin-mitochondrion connection along the evolution, and the role of melatonin in mitochondrial homeostasis. | |||
# [http://www.ncbi.nlm.nih.gov/pubmed/21244359 Acuna Castroviejo D, Lopez LC, Escames G, Lopez A, Garcia JA, Reiter RJ (2011) Melatonin-mitochondria interplay in health and disease. Curr Top Med Chem 11: 221-240.] | |||
Tan | # [http://www.ncbi.nlm.nih.gov/pubmed?term=Mitochondria%20and%20chloroplasts%20as%20the%20original%20sites%20of%20melatonin%20synthesis%3A%20a%20hypothesis%20related%20to%20melatonin's%20primary%20function%20and%20evolution%20in%20eukaryotes Tan DX, Manchester LC, Liu X, Rosales-Corral SA, Acuna-Castroviejo D, Reiter RJ (2012) Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin's primary function and evolution in eukaryotes. J Pineal Res. doi: 10.1111/jpi.12026] | ||
|keywords=Melatonin, Mitochondria, Reactive oxygen species, Respiratory chain, ATP | |||
|mipnetlab=ES Granada Acuna-Castroviejo D | |||
|keywords= | |||
|journal=Mitochondr Physiol Network | |journal=Mitochondr Physiol Network | ||
|articletype=Abstract | |articletype=Abstract | ||
}} | }} | ||
{{Labeling | {{Labeling | ||
| | |area=Respiration, mt-Biogenesis;mt-density, mt-Structure;fission;fusion, mt-Membrane, mt-Medicine | ||
|organism=Mouse, Rat | |organism=Mouse, Rat | ||
|tissues= | |tissues=Heart, Skeletal muscle, Nervous system, Liver | ||
|preparations=Intact | |preparations=Intact organism, Intact cells, Isolated mitochondria | ||
|enzymes=Complex I, Complex II;succinate dehydrogenase, Complex III, Complex V;ATP synthase | |||
|injuries=Oxidative stress;RONS | |||
|diseases=Aging;senescence, Alzheimer's, Cancer, Neurodegenerative, Parkinson's, Sepsis | |||
|topics=ATP production, Ion;substrate transport, mt-Membrane potential, Redox state | |||
|couplingstates=ROUTINE, OXPHOS | |couplingstates=ROUTINE, OXPHOS | ||
| | |pathways=N, S, NS | ||
| | |instruments=Oxygraph-2k | ||
|journal=Mitochondr Physiol Network | |journal=Mitochondr Physiol Network | ||
|articletype=Abstract | |articletype=Abstract | ||
| Line 34: | Line 34: | ||
== Affiliations and author contributions == | == Affiliations and author contributions == | ||
Instituto de Biotecnología, Centro de Investigación Biomédica, Parque Tecnológico de Ciencias de la Salud, Universidad de | Instituto de Biotecnología, Centro de Investigación Biomédica, Parque Tecnológico de Ciencias de la Salud, Universidad de Granada, Spain. - [email protected] | ||
Supported by grants # P10-CTS-5784 and PI08-1664. | |||
== Figure 1 == | == Figure 1 == | ||
[[File:Figura.JPG|900px|Melatonin and mitochondria]] | |||
== Help == | == Help == | ||
* [[MitoPedia: Terms and abbreviations]] | |||
* [[MitoPedia | |||
Latest revision as of 15:44, 7 November 2016
| Acuna-Castroviejo D (2012) Melatonin and the quality of life: the melatonin-mitochondrion connection. Mitochondr Physiol Network 17.12. |
Link: MiPNet17.12 Bioblast 2012 - Open Access
Acuna-Castroviejo D (2012)
Event: Bioblast 2012
The first relationship between melatonin and mitochondria came from histological studies showing changes in mitochondrial density and morphology after pinealectomy or melatonin administration to experimental animals. After the discovery of the antioxidant activity of melatonin in 1993, the possibility that melatonin exerts its effects on the mitochondria, the main ROS-producing organelle, was hypothesized. The first experiments demonstrated a highly efficient ability of melatonin to counteract the mitochondrial oxidative stress in vitro and in vivo. In parallel, melatonin increases the respiratory chain activity, reduces the oxygen consumption, and increases the ATP production. In some of these experiments we could demonstrated that the mitochondria take up melatonin in a time- and concentration-dependent manner. To further analyze the ability of melatonin to prevent and/or counteract mitochondrial dysfunction, different experimental models of aging and disease, including sepsis, Parkinson’s disease, and Alzheimer’s disease, was evaluated. In all of them, melatonin administration restored the full bioenergetic capacity of the mitochondria, restoring or even increasing their ATP production. Along this time, it was shown that most of the tissues and organs produce melatonin independently of the pineal gland. An important feature of this extrapineal source of melatonin is that its synthetizing enzymes, AANAT and ASMT, are inducible, i.e., the cells produce melatonin when they require it for protective purposes. This melatonin does not exit to the extracellular fluid. To further analyze the dynamics of the extrapineal melatonin, we recently studied the subcellular distribution of the indoleamine in liver and brain. These studies showed that melatonin is produced in considerably higher amounts in these tissues than in the pineal gland, it is not uniformly distributed in the cell, and is mainly located in the membrane, mitochondria and nucleus. Interestingly, membrane content of melatonin increased in a dose-dependent manner after administration of melatonin to rats, but the content of the indoleamine in the nucleus and mitochondria is saturated. There are now evidences of the ability of mitochondria (and chloroplasts) to synthesize melatonin, which explains the high levels of the indoleamine in this organelle. The phylogenetic origins of the mitochondria, and the presence of melatonin in ancient one cell organisms, speak in favor of a melatonin-mitochondrion connection along the evolution, and the role of melatonin in mitochondrial homeostasis.
- Acuna Castroviejo D, Lopez LC, Escames G, Lopez A, Garcia JA, Reiter RJ (2011) Melatonin-mitochondria interplay in health and disease. Curr Top Med Chem 11: 221-240.
- Tan DX, Manchester LC, Liu X, Rosales-Corral SA, Acuna-Castroviejo D, Reiter RJ (2012) Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin's primary function and evolution in eukaryotes. J Pineal Res. doi: 10.1111/jpi.12026
• Keywords: Melatonin, Mitochondria, Reactive oxygen species, Respiratory chain, ATP
• O2k-Network Lab: ES Granada Acuna-Castroviejo D
Labels: MiParea: Respiration, mt-Biogenesis;mt-density, mt-Structure;fission;fusion, mt-Membrane, mt-Medicine Pathology: Aging;senescence, Alzheimer's, Cancer, Neurodegenerative, Parkinson's, Sepsis Stress:Oxidative stress;RONS Organism: Mouse, Rat Tissue;cell: Heart, Skeletal muscle, Nervous system, Liver Preparation: Intact organism, Intact cells, Isolated mitochondria Enzyme: Complex I, Complex II;succinate dehydrogenase, Complex III, Complex V;ATP synthase Regulation: ATP production, Ion;substrate transport, mt-Membrane potential, Redox state Coupling state: ROUTINE, OXPHOS Pathway: N, S, NS HRR: Oxygraph-2k
Affiliations and author contributions
Instituto de Biotecnología, Centro de Investigación Biomédica, Parque Tecnológico de Ciencias de la Salud, Universidad de Granada, Spain. - [email protected]
Supported by grants # P10-CTS-5784 and PI08-1664.