Difference between revisions of "Fischer 2021 MitoFit Fe liver"
| (5 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
{{MitoFit page name}} | {{MitoFit page name}} | ||
{{Publication | {{Publication | ||
|title=Fischer C, Volani C, Komlódi T, Seifert M, Demetz E, Valente de Souza L, Auer K, Petzer V, von Raffay L, Moser P, Gnaiger E, Weiss G (2021) Dietary iron overload and ''Hfe<sup>-/-</sup>'' related hemochromatosis alter hepatic mitochondrial function. | |title=Fischer C, Volani C, Komlódi T, Seifert M, Demetz E, Valente de Souza L, Auer K, Petzer V, von Raffay L, Moser P, Gnaiger E, Weiss G (2021) Dietary iron overload and ''Hfe<sup>-/-</sup>'' related hemochromatosis alter hepatic mitochondrial function. https://doi.org/10.26124/mitofit:2021-0009 - ''2021-11-16 Published in [[Fischer 2021 Antioxidants |»Antioxidants«]]'' | ||
|info=[[File:MitoFit Preprints pdf.png|left|160px|link=https://wiki.oroboros.at/images/b/bd/Fischer_2021_MitoFit_Fe_liver.pdf |MitoFit pdf]] [https://wiki.oroboros.at/images/b/bd/Fischer_2021_MitoFit_Fe_liver.pdf Dietary iron overload and ''Hfe<sup>-/-</sup>'' related hemochromatosis alter hepatic mitochondrial function] | |info=MitoFit Preprints 2021.9. [[File:MitoFit Preprints pdf.png|left|160px|link=https://wiki.oroboros.at/images/b/bd/Fischer_2021_MitoFit_Fe_liver.pdf |MitoFit pdf]] [https://wiki.oroboros.at/images/b/bd/Fischer_2021_MitoFit_Fe_liver.pdf Dietary iron overload and ''Hfe<sup>-/-</sup>'' related hemochromatosis alter hepatic mitochondrial function] | ||
|authors= | |authors=MitoFit Preprints | ||
|year=2021-10-07 | |year=2021-10-07 | ||
|journal=MitoFit Prep | |journal=MitoFit Prep | ||
|abstract= | |abstract='''Journal publication 2021-11-16 in [[Fischer 2021 Antioxidants |»Antioxidants«]]'''</big> | ||
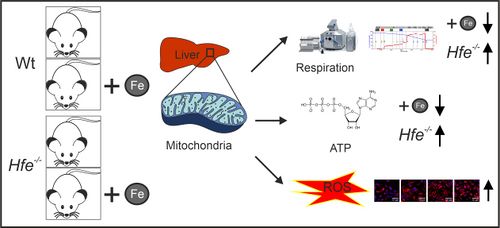
[[File:Fischer_2021_MitoFit_Fe_liver - graphical abstract.png|right|500px|Graphical abstract]] Iron is an essential co-factor for many cellular metabolic processes, and mitochondria are main sites of utilization. Iron accumulation promotes production of reactive oxygen species (ROS) via the catalytic activity of iron species. Herein, we investigated the consequences of dietary and genetic iron overload on mitochondrial function. C57/BL6N wildtype and ''Hfe<sup>-/-</sup>'' mice, the latter a genetic hemochromatosis model, received either normal diet (ND) or high iron diet (HI) for two weeks. Liver mitochondrial respiration was measured using high-resolution respirometry along with analysis of expression of specific proteins and ROS production. HI promoted tissue iron accumulation and slightly affected mitochondrial function in wildtype mice. Hepatic mitochondrial function was impaired in ''Hfe<sup>-/-</sup>'' mice on ND and HI. Compared to wildtype mice, ''Hfe<sup>-/-</sup>'' mice on ND showed increased mitochondrial respiratory capacity. ''Hfe<sup>-/-</sup>'' mice on HI showed very high liver iron levels, decreased mitochondrial respiratory capacity and increased ROS production associated with reduced mitochondrial aconitase activity. Although ''Hfe<sup>-/-</sup>'' resulted in increased mitochondrial iron loading, the concentration of metabolically reactive cytoplasmic iron and mitochondrial density remained unchanged. Our data shows multiple effects of dietary and genetic iron loading on mitochondrial function and linked metabolic pathways, providing an explanation for fatigue in iron-overloaded hemochromatosis patients and suggests iron reduction therapy for improvement of mitochondrial function. | [[File:Fischer_2021_MitoFit_Fe_liver - graphical abstract.png|right|500px|Graphical abstract]] Iron is an essential co-factor for many cellular metabolic processes, and mitochondria are main sites of utilization. Iron accumulation promotes production of reactive oxygen species (ROS) via the catalytic activity of iron species. Herein, we investigated the consequences of dietary and genetic iron overload on mitochondrial function. C57/BL6N wildtype and ''Hfe<sup>-/-</sup>'' mice, the latter a genetic hemochromatosis model, received either normal diet (ND) or high iron diet (HI) for two weeks. Liver mitochondrial respiration was measured using high-resolution respirometry along with analysis of expression of specific proteins and ROS production. HI promoted tissue iron accumulation and slightly affected mitochondrial function in wildtype mice. Hepatic mitochondrial function was impaired in ''Hfe<sup>-/-</sup>'' mice on ND and HI. Compared to wildtype mice, ''Hfe<sup>-/-</sup>'' mice on ND showed increased mitochondrial respiratory capacity. ''Hfe<sup>-/-</sup>'' mice on HI showed very high liver iron levels, decreased mitochondrial respiratory capacity and increased ROS production associated with reduced mitochondrial aconitase activity. Although ''Hfe<sup>-/-</sup>'' resulted in increased mitochondrial iron loading, the concentration of metabolically reactive cytoplasmic iron and mitochondrial density remained unchanged. Our data shows multiple effects of dietary and genetic iron loading on mitochondrial function and linked metabolic pathways, providing an explanation for fatigue in iron-overloaded hemochromatosis patients and suggests iron reduction therapy for improvement of mitochondrial function. | ||
| Line 13: | Line 13: | ||
|keywords=hemochromatosis; iron overload; reactive oxygen species, ROS; mitochondria, mt; mitochondrial respiration; liver; fatigue | |keywords=hemochromatosis; iron overload; reactive oxygen species, ROS; mitochondria, mt; mitochondrial respiration; liver; fatigue | ||
|editor=Cardoso LHD | |editor=Cardoso LHD | ||
}} | }} | ||
ORC'''ID''': [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-5656-5030]] [[Fischer C]], [[File:ORCID.png|20px|link=https://orcid.org/0000-0003-3600-4735]] [[Volani C]], [[File:ORCID.png|20px|link=https://orcid.org/0000-0001-9876-1411]] [[Komlodi T]], [[Seifert M]], [[Demetz E]], [[File:ORCID.png|20px|link=https://orcid.org/0000-0002-0370-2791]] [[Valente de Souza L]], [[Auer K]], [[Petzer V]], [[von Raffay L]], [[Moser P]], [[File:ORCID.png|20px|link=https://orcid.org/0000-0003-3647-5895]] [[Gnaiger E]], [[File:ORCID.png|20px|link=https://orcid.org/0000-0003-0709-2158]] [[Weiss G]] | |||
Revision as of 16:31, 4 June 2022
Fischer 2021 MitoFit Fe liver
| Fischer C, Volani C, Komlódi T, Seifert M, Demetz E, Valente de Souza L, Auer K, Petzer V, von Raffay L, Moser P, Gnaiger E, Weiss G (2021) Dietary iron overload and Hfe-/- related hemochromatosis alter hepatic mitochondrial function. https://doi.org/10.26124/mitofit:2021-0009 - 2021-11-16 Published in »Antioxidants« |
» MitoFit Preprints 2021.9.
Dietary iron overload and Hfe-/- related hemochromatosis alter hepatic mitochondrial function
MitoFit Preprints (2021-10-07) MitoFit Prep
Abstract: Journal publication 2021-11-16 in »Antioxidants«
Iron is an essential co-factor for many cellular metabolic processes, and mitochondria are main sites of utilization. Iron accumulation promotes production of reactive oxygen species (ROS) via the catalytic activity of iron species. Herein, we investigated the consequences of dietary and genetic iron overload on mitochondrial function. C57/BL6N wildtype and Hfe-/- mice, the latter a genetic hemochromatosis model, received either normal diet (ND) or high iron diet (HI) for two weeks. Liver mitochondrial respiration was measured using high-resolution respirometry along with analysis of expression of specific proteins and ROS production. HI promoted tissue iron accumulation and slightly affected mitochondrial function in wildtype mice. Hepatic mitochondrial function was impaired in Hfe-/- mice on ND and HI. Compared to wildtype mice, Hfe-/- mice on ND showed increased mitochondrial respiratory capacity. Hfe-/- mice on HI showed very high liver iron levels, decreased mitochondrial respiratory capacity and increased ROS production associated with reduced mitochondrial aconitase activity. Although Hfe-/- resulted in increased mitochondrial iron loading, the concentration of metabolically reactive cytoplasmic iron and mitochondrial density remained unchanged. Our data shows multiple effects of dietary and genetic iron loading on mitochondrial function and linked metabolic pathways, providing an explanation for fatigue in iron-overloaded hemochromatosis patients and suggests iron reduction therapy for improvement of mitochondrial function.
• Keywords: hemochromatosis; iron overload; reactive oxygen species, ROS; mitochondria, mt; mitochondrial respiration; liver; fatigue
• Bioblast editor: Cardoso LHD
ORCID: ![]() Fischer C,
Fischer C, ![]() Volani C,
Volani C, ![]() Komlodi T, Seifert M, Demetz E,
Komlodi T, Seifert M, Demetz E, ![]() Valente de Souza L, Auer K, Petzer V, von Raffay L, Moser P,
Valente de Souza L, Auer K, Petzer V, von Raffay L, Moser P, ![]() Gnaiger E,
Gnaiger E, ![]() Weiss G
Weiss G