Description
Abbreviation: FNS(Oct,GM)
Reference: A: ![]() »Versions
»Versions
- SUIT-category: FNS(Oct,GM)
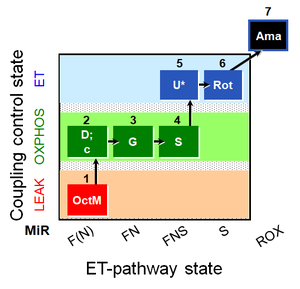
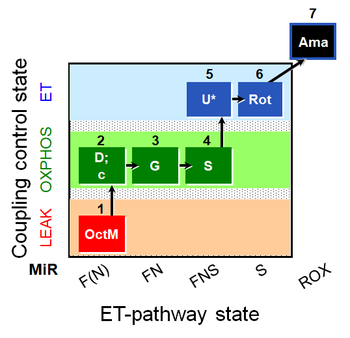
- SUIT protocol pattern: diametral 1OctM;2D;3G;4S;5U;6Rot;7Ama
DLP applications
References
| Year | Reference | Organism | Tissue;cell | |
|---|---|---|---|---|
| Pesta 2012 Methods Mol Biol | 2012 | Pesta D, Gnaiger E (2012) High-resolution respirometry. OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods Mol Biol 810:25-58. https://doi.org/10.1007/978-1-61779-382-0_3 | Human | Skeletal muscle Other cell lines HEK Fibroblast HUVEC |
Steps and respiratory states
| Step | State | Pathway | Q-junction | Comment - Events (E) and Marks (M) |
|---|---|---|---|---|
| 1OctM | OctML(n) | F(N) | FAO | 1OctM
Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. Low concentration of malate, typically 0.1 mM, does not saturate the N-pathway; but saturates the F-pathway. Non-phosphorylating resting state (LEAK state); LEAK respiration L(n) in the absence of ADP, ATP, AMP (no adenylates). |
| 2D | OctMP | F(N) | FAO | 1OctM;2D
Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. Low concentration of malate, typically 0.1 mM, does not saturate the N-pathway; but saturates the F-pathway. OXPHOS capacity P (with saturating [ADP]), active OXPHOS state. |
| 2c | OctMP | F(N) | FAO | 1OctM;2D;2c
Addition of cytochrome c yields a test for integrity of the mtOM (cytochrome c control efficiency). Stimulation by added cytochrome c would indicate an injury of the mtOM and limitation of respiration in the preceding state without added c due to loss of cytochrome c. Typically, cytochrome c is added immediately after the earliest ADP-activation step (OXPHOS capacity P with saturating [ADP]).Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. Low concentration of malate, typically 0.1 mM, does not saturate the N-pathway; but saturates the F-pathway. OXPHOS capacity P (with saturating [ADP]), active OXPHOS state. |
| 3G | OctGMP | FN | F&CI | 1OctM;2D;2c;3G
NADH-linked substrates (type N-pathway to Q). Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. OXPHOS capacity P (with saturating [ADP]), active OXPHOS state. |
| 4S | OctGMSP | FNS | F&CI&II | 1OctM;2D;2c;3G;4S
Respiratory stimulation by simultaneous action of type N substrates & succinate, with convergent electron flow in the NS-pathway for reconstitution of TCA cycle function. Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. OXPHOS capacity P (with saturating [ADP]), active OXPHOS state. |
| 5U | OctGMSE | FNS | F&CI&II | 1OctM;2D;2c;3G;4S;5U
Respiratory stimulation by simultaneous action of type N substrates & succinate, with convergent electron flow in the NS-pathway for reconstitution of TCA cycle function. Respiratory stimulation of the FAO-pathway, F, by fatty acid, FA, in the presence of malate, M. Malate is a type N substrate (N), required for the F-pathway. The FA concentration has to be optimized to saturate the F-pathway, without inhibiting or uncoupling respiration. Noncoupled electron transfer state, ET state, with ET capacity E. |
| 6Rot | SE | S | CII | 1OctM;2D;2c;3G;4S;5U;6Rot
Succinate pathway control state (S-pathway) after inhibiting CI with rotenone, which also inhibits the F-pathway. Noncoupled electron transfer state, ET state, with ET capacity E. |
| 7Ama | ROX | 1OctM;2D;2c;3G;4S;5U;6Rot;7Ama
Rox is the residual oxygen consumption in the ROX state, due to oxidative side reactions, estimated either after inhibition of CIII (e.g. antimycin A, myxothiazol), CIV (e.g. Cyanide) or in the absence of endogenous fuel-substrates. Rox is subtracted from oxygen flux as a baseline for all respiratory states, to obtain mitochondrial respiration. |
| Step | Respiratory state | Pathway control | ET-Complex | Comment |
|---|---|---|---|---|
| ## AsTm | AsTmE | CIV | CIV | |
| ## Azd | CHB |
- Bioblast links: SUIT protocols - >>>>>>> - Click on [Expand] or [Collapse] - >>>>>>>
- Coupling control
- Pathway control
- Main fuel substrates
- » Glutamate, G
- » Glycerophosphate, Gp
- » Malate, M
- » Octanoylcarnitine, Oct
- » Pyruvate, P
- » Succinate, S
- Main fuel substrates
- Glossary
Strenghts and limitations
- Comparison of GM- with PM-capacity yields important information on N-pathway respiratory control upstream of CI (Lemeux et al 2017; Votion et al 2012).
- + Glutamate is easier to prepare compared to pyruvate.
- + Reasonable duration of the experiment.
Compare SUIT protocols
MitoPedia concepts:
MiP concept,
SUIT protocol,
Recommended
MitoPedia methods:
Respirometry